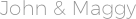The aqueous phase is characterized by a constant ionic strength of 4.0 due to the salt background of sodium perchlorate and salts of NaBr, NaJ or NaSCN of variable concentrations. The likelihood of the formation of perchlorate and mixed-ligand complexes also applies to extraction systems based on 4-methyl-2-pentanone and 4-methyl-2-pentanol, the extractable forms of which are compounds of the composition, ,, where bromine, iodide, or thiocyanate ion is present as the halide and pseudohalide of the ligand. In the aqueous phase, there are only chloride complexes, which are subjected to extraction with various oxygen-containing solvents.Įxtraction of III subgroup metal ions with di (2-ethylhexyl) phosphoric acid depending on the concentration of perchloric acid: 1 - Ga (III), 2 - In (III), 3 - Tl (III).

The latter formation is possibly caused by the coextractability of perchloric acid due to its higher extraction affinity as compared to hydrochloric acid. It is also assumed that during the extraction of indium in aqueous solutions of hydrochloric acid at its various concentrations and constant ionic strength maintained by the addition of perchloric acid, indium is present in the organic phase in the form of ionic aggregates HClO 4⋅HInCl 4. At the same time, from concentrated solutions of perchloric acid for cationic reagents - alkylphosphoric acids (for example, di-2-ethylhexylphosphoric acid Figure 1) - a possible extractable form of indium compounds is the complex cation 2+. The absence of complex compounds of indium with perchlorate ion is also indicated by the data of liquid extraction, in particular, three n-octylamine does not extract indium from a solution with an HClO 4 concentration less than 2 M. It was not possible to detect the binding of indium to perchlorate ions ClO 4 − by special experiments carried out in mixed water-non-aqueous mixtures and organic media. The proton-magnetic resonance measurement of the coordination number of perchlorate water-organic systems testifies to the six fold coordination of water molecules around the indium cation and to the absence of strong evidence of contact ion pairing. Similar results, denying the likelihood of coordination interaction of In 3+ with ClO 4 −, were obtained when studying the solvation of cations by NMR on H 1 and P 31 nuclei in aqueous-organic mixtures. Also, no intrinsic complexation influencing the activity of In 3+ was reliably detected up to a sodium perchlorate concentration of 16 mol/kg and with an In(ClO 4) 3 content of more than 4 M. Spectrophotometrically in the ultraviolet region of the spectrum, there were no significant signs of direct interaction in aqueous solutions of In(ClO 4) 3 salts. There is no evidence of the complexing effect of the highly concentrated electrolyte NaClO 4 1.1÷9.1 M and by electrophoresis. According to the data of the Raman spectra, the aquocomplex is stable in acidic perchlorate solutions, and in the studied concentration range, neither inner sphere nor hydroxostructures are formed. Raman and IR spectroscopic studies of indium hydration in perchlorate solutions revealed octahedral hexaaquocation 3 +. In the Raman spectra of the perchlorate solution, only the lines of the ClO 4 − anion and the aquocomplexes of the In 3 + ion ⋅aq were found. However, partial formation of ion pairs between them is allowed. IR spectra including near infrared and spectroscopy of disturbed total internal reflection, NMR signals. The absence of coordination interaction of acid ions in the “In 3 + - ClO 4 −– H 2O” system is indicated by the spectra of Raman scattering. These fairly well known provisions are fully confirmed for indium perchlorate, which is proved by various research methods. The successful synthesis of perchloro-complexes in work is associated with the use of anhydrous perchloric acid as a reaction medium. The formation of a coordination bond between the perchlorate ion and the complexing cation is possible only in acceptor or very weakly donating solvents. Water molecules and most non-aqueous donor media displace such a weak ligand as the ClO 4 − ion from the inner sphere of the compound. The difficulty in preparing perchlorate metal complexes is the lack of suitable solvents.
It is known that perchlorate ion does not form complex compounds even with extremely strong complexing metal ions. Ionic state of indium in perchlorate solutions